10 mg
Prices exclude VAT
This product has been discontinued
| Content: | 10 mg or 50 mg |
| Shipping Temperature: | Ambient |
| Storage Temperature: | Below -10oC |
| Physical Form: | Solid |
| Stability: | > 10 years under recommended storage conditions |
| CAS Number: | 6819-07-04 |
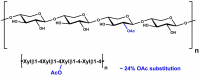
| Molecular Formula: | C16H21NO11 |
| Molecular Weight: | 403.3 |
| Purity: | > 97% |
| Substrate For (Enzyme): | β-Xylosidase |
| Assay Format: | Spectrophotometer, Microplate, Auto-analyser |
| Detection Method: | Absorbance |
| Wavelength (nm): | 400-420 |
These products have been discontinued (read more).
High purity 4-Nitrophenyl-β-xylobioside for use in research, biochemical enzyme assays and in vitro diagnostic analysis.
This is a colourimetric substrate for measurement of β-xylanase and β-xylosidase activity.
Data booklets for each pack size are located in the Documents tab.
Mangan, D., Cornaggia, C., Liadova, A., McCormack, N., Ivory, R., McKie, V. A., Ormerod, A. & McCleary, D. V. (2017). Carbohydrate Research, 445, 14-22.
endo-1,4-β-Xylanase (EC 3.2.1.8) is employed across a broad range of industries including animal feed, brewing, baking, biofuels, detergents and pulp (paper). Despite its importance, a rapid, reliable, reproducible, automatable assay for this enzyme that is based on the use of a chemically defined substrate has not been described to date. Reported herein is a new enzyme coupled assay procedure, termed the XylX6 assay, that employs a novel substrate, namely 4,6-O-(3-ketobutylidene)-4-nitrophenyl-β-45-O-glucosyl-xylopentaoside. The development of the substrate and associated assay is discussed here and the relationship between the activity values obtained with the XylX6 assay versus traditional reducing sugar assays and its specificity and reproducibility were thoroughly investigated.
Hide AbstractVersatile high resolution oligosaccharide microarrays for plant glycobiology and cell wall research.
Pedersen, H. L., Fangel, J. U., McCleary, B., Ruzanski, C., Rydahl, M. G., Ralet, M. C., Farkas, V., Von Schantz, L., Marcus, S. E., Andersen, M.C. F., Field, R., Ohlin, M., Knox, J. P., Clausen, M. H. & Willats, W. G. T. (2012). Journal of Biological Chemistry, 287(47), 39429-39438.
Microarrays are powerful tools for high throughput analysis, and hundreds or thousands of molecular interactions can be assessed simultaneously using very small amounts of analytes. Nucleotide microarrays are well established in plant research, but carbohydrate microarrays are much less established, and one reason for this is a lack of suitable glycans with which to populate arrays. Polysaccharide microarrays are relatively easy to produce because of the ease of immobilizing large polymers noncovalently onto a variety of microarray surfaces, but they lack analytical resolution because polysaccharides often contain multiple distinct carbohydrate substructures. Microarrays of defined oligosaccharides potentially overcome this problem but are harder to produce because oligosaccharides usually require coupling prior to immobilization. We have assembled a library of well characterized plant oligosaccharides produced either by partial hydrolysis from polysaccharides or by de novo chemical synthesis. Once coupled to protein, these neoglycoconjugates are versatile reagents that can be printed as microarrays onto a variety of slide types and membranes. We show that these microarrays are suitable for the high throughput characterization of the recognition capabilities of monoclonal antibodies, carbohydrate-binding modules, and other oligosaccharide-binding proteins of biological significance and also that they have potential for the characterization of carbohydrate-active enzymes.
Hide AbstractAssessing and enhancing foldability in designed proteins.
Fleishman, S. J., Listov, D., Lipsh-Sokolik, R., Yang, C. & Correia, B. E. (2021). BioRxiv, In Press.
Recent advances in protein-design methodology have led to a dramatic increase in its reliability and scale. With these advances, dozens and even thousands of designed proteins are automatically generated and screened. Nevertheless, the success rate, particularly in design of functional proteins, is low and fundamental goals such as reliable de novo design of efficient enzymes remain beyond reach. Experimental analyses have consistently indicated that a major cause of design failure is inaccuracy and misfolding relative to the design model. To address this challenge, we describe complementary methods to diagnose and ameliorate suboptimal regions in designed proteins: first, we develop a Rosetta atomistic computational mutation scanning approach to detect energetically suboptimal positions in designs; second, we demonstrate that the AlphaFold2 ab initio structure prediction method flags regions that may misfold in designed enzymes and binders; and third, we focus FuncLib design calculations on suboptimal positions in a previously designed low-efficiency enzyme, thereby improving its catalytic efficiency by 330 fold. Thus, foldability analysis and enhancement may dramatically increase the success rate in design of functional proteins.
Hide AbstractProduction of xylanolytic enzymes by Moesziomyces spp. using xylose, xylan and brewery’s spent grain as substrates.
Faria, N. T., Marques, S., Ferreira, F. C. & Fonseca, C. (2019). New Biotechnology, 49, 137-143.
Xylanases play a crucial role in the hydrolysis of xylan-rich hemicelluloses and have wide industrial applications in the fuel, food, feed and pulp and paper industries. The production of these enzymes at low cost is of paramount importance for their commercial deployment. Moesziomyces antarcticus PYCC 5048T and M. aphidis PYCC 5535T were screened for their ability to produce xylanolytic enzymes when grown on d-xylose, xylan (beechwood) and brewery’s spent grain (BSG). The extracellular crude extracts produced were characterized and tested in xylan hydrolysis. The yeasts produced xylanolytic enzymes without cellulolytic activity on all the substrates tested. The highest xylanase volumetric activity was obtained with M. aphidis PYCC 5535T grown on BSG, reaching 518.2 U/ml, a value 8.4- and 4.7-fold higher than those achieved on xylan and d-xylose, respectively. The xylanase activities were characterized in relation to pH and temperature with optima at 4.5 and 50°C, respectively. The extracts from both M. antarcticus PYCC 5048Tand M. aphidis PYCC 5535T were used in xylan hydrolysis, producing d-xylose as the major end product (0.43 and 0.34-0.47 gD-xylose/gxylan, respectively, at 50°C) and relatively low or no xylobiose accumulation (from no detection to 0.12 gD-xylobiose/gxylan at 50°C).
Hide AbstractPell, G., Taylor, E. J., Gloster, T. M., Turkenburg, J. P., Fontes, C. M. G. A., Ferreira, L. M., Nagy, T., Clark, S. J., Davies, G. J. & Gilbert, H. J. (2004). Journal of Biological Chemistry, 279(10), 9597-9605.
Endo-β-1,4-xylanases (xylanases), which cleave β-1,4 glycosidic bonds in the xylan backbone, are important components of the repertoire of enzymes that catalyze plant cell wall degradation. The mechanism by which these enzymes are able to hydrolyze a range of decorated xylans remains unclear. Here we reveal the three-dimensional structure, determined by x-ray crystallography, and the catalytic properties of the Cellvibrio mixtus enzyme Xyn10B (CmXyn10B), the most active GH10 xylanases described to date. The crystal structure of the enzyme in complex with xylopentaose reveals that at the +1 subsite the xylose moiety is sandwiched between hydrophobic residues, which is likely to mediate tighter binding than in other GH10 xylanases. The crystal structure of the xylanase in complex with a range of decorated xylooligosaccharides reveals how this enzyme is able to hydrolyze substituted xylan. Solvent exposure of the O-2 groups of xylose at the +4, +3, +1, and -3 subsites may allow accommodation of the α-1,2-linked 4-O-methyl-D-glucuronic acid side chain in glucuronoxylan at these locations. Furthermore, the uronic acid makes hydrogen bonds and hydrophobic interactions with the enzyme at the +1 subsite, indicating that the sugar decorations in glucuronoxylan are targeted to this proximal aglycone binding site. Accommodation of 3′-linked L-arabinofuranoside decorations is observed in the -2 subsite and could, most likely, be tolerated when bound to xylosides in -3 and +4. A notable feature of the binding mode of decorated substrates is the way in which the subsite specificities are tailored both to prevent the formation of “dead-end” reaction products and to facilitate synergy with the xylan degradation-accessory enzymes such as α-glucuronidase. The data described in this report and in the accompanying paper (Fujimoto, Z., Kaneko, S., Kuno, A., Kobayashi, H., Kusakabe, I., and Mizuno, H. (2004) J. Biol. Chem. 279, 9606-9614) indicate that the complementarity in the binding of decorated substrates between the glycone and aglycone regions appears to be a conserved feature of GH10 xylanas.
Hide Abstract