110 assays (manual) / 1100 assays (microplate) / 1100 assays (auto-analyser)
Prices exclude VAT
Available for shipping
| Content: | 110 assays (manual) / 1100 assays (microplate) / 1100 assays (auto-analyser) |
| Shipping Temperature: | Ambient |
| Storage Temperature: | Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
| Analyte: | D-Fructose, D-Glucose |
| Assay Format: | Spectrophotometer, Microplate, Auto-analyser |
| Detection Method: | Absorbance |
| Wavelength (nm): | 340 |
| Signal Response: | Increase |
| Linear Range: | 4 to 80 µg of D-glucose, D-fructose or sucrose per assay |
| Limit of Detection: | 0.66 mg/L |
| Reaction Time (min): | ~ 13 min |
| Application examples: | Wine, beer, fruit juices, soft drinks, milk, jam, honey, dietetic foods, bread, bakery products, candies, desserts, confectionery, ice-cream, fruit and vegetables, condiments, tobacco, cosmetics, pharmaceuticals, paper and other materials (e.g. biological cultures, samples, etc.). |
| Method recognition: | Methods based on this principle have been accepted by AOAC Method 985.09, EN, NEN, NF, DIN, GOST, OIV, IFU, AIJN, MEBAK and IOCCC |
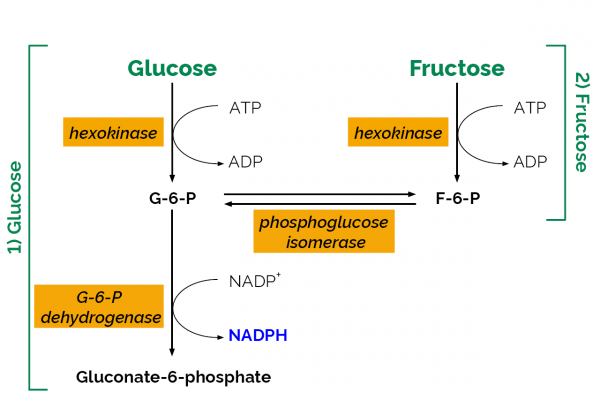
D-Fructose/D-Glucose test kit, an enzymatic UV-method for the measurement and analysis of D-fructose and/or D-glucose in plant and food products.
Note for Content: The number of manual tests per kit can be doubled if all volumes are halved. This can be readily accommodated using the MegaQuantTM Wave Spectrophotometer (D-MQWAVE).
See more of our monosaccharide assay kits.

- Extended cofactors stability. Dissolved cofactors stable for > 1 year at 4oC.
- PVP incorporated to prevent tannin inhibition
- Validated by the University of Wine, Suze la Rousse, France
- Very competitive price (cost per test)
- All reagents stable for > 2 years after preparation (manual analysis applications)
- Rapid reaction at either 25 or 37oC
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
- Suitable for manual, microplate and auto-analyser formats
Megazyme “advanced” wine test kits general characteristics and validation.
Charnock, S. J., McCleary, B. V., Daverede, C. & Gallant, P. (2006). Reveue des Oenologues, 120, 1-5.
Many of the enzymatic test kits are official methods of prestigious organisations such as the Association of Official Analytical Chemicals (AOAC) and the American Association of Cereal Chemists (AACC) in response to the interest from oenologists. Megazyme decided to use its long history of enzymatic bio-analysis to make a significant contribution to the wine industry, by the development of a range of advanced enzymatic test kits. This task has now been successfully completed through the strategic and comprehensive process of identifying limitations of existing enzymatic bio-analysis test kits where they occurred, and then using advanced techniques, such as molecular biology (photo 1), to rapidly overcome them. Novel test kits have also been developed for analytes of emerging interest to the oenologist, such as yeast available nitrogen (YAN; see pages 2-3 of issue 117 article), or where previously enzymes were simply either not available, or were too expensive to employ, such as for D-mannitol analysis.
Hide AbstractGrape and wine analysis: Oenologists to exploit advanced test kits.
Charnock, S. C. & McCleary, B. V. (2005). Revue des Enology, 117, 1-5.
It is without doubt that testing plays a pivotal role throughout the whole of the vinification process. To produce the best possible quality wine and to minimise process problems such as “stuck” fermentation or troublesome infections, it is now recognised that if possible testing should begin prior to harvesting of the grapes and continue through to bottling. Traditional methods of wine analysis are often expensive, time consuming, require either elaborate equipment or specialist expertise and frequently lack accuracy. However, enzymatic bio-analysis enables the accurate measurement of the vast majority of analytes of interest to the wine maker, using just one piece of apparatus, the spectrophotometer (see previous issue No. 116 for a detailed technical review). Grape juice and wine are amenable to enzymatic testing as being liquids they are homogenous, easy to manipulate, and can generally be analysed without any sample preparation.
Hide AbstractInfluence of Post-Harvest 1-Methylcyclopropene (1-MCP) Treatment and Refrigeration on Chemical Composition, Phenolic Profile and Antioxidant Modifications during Storage of Abate Fétel Pears.
Tedeschi, P., Marzocchi, S., Marchetti, N., Barba, F. J. & Maietti, A. (2023). Antioxidants, 12(11), 1955.
‘Abate Fétel’, a winter cultivar, is the most important pear cultivar in Italy; its fruits are appreciated by consumers for their aroma, texture and balanced sweet and sour taste. Maintaining high-quality characteristics to prolong the shelf-life of fruit and preserve the sensory and nutritional quality is a priority for the food industry. The aim of our study was to test the effectiveness of 1-methylcyclopropene (1-MCP) and cold storage in prolonging the shelf-life of these fruits, which were harvested at maturity at two different times. This work focused on the effects of different storage treatments and two ripening times on (i) the chemical composition of Abate Fétel pulp fruits to preserve their sweet taste and aroma and (ii) the phenolic profile composition and antioxidant activity of the peel, which is naturally rich in phytochemicals and important for the fruit’s shelf-life and in the functional food industry for its high nutritional value. Abate Fétel fruits were harvested at the optimal commercial maturity stage, first on 15 September, having been treated with 1-MCP and stored for 2 months at cold temperatures; the other fruits were harvested at the end of September and stored in a cold cell for 2 months. The fruit pulp was tested for glucose and fructose, pH, acidity and organic acids (malic, citric, fumaric and shikimic), phenolic content and phenolic compounds (chlorogenic and caffeic acids, rutin, hyperoside, kaempferol-3-rutinoside and isoquercitrin), and the antioxidant activities in the fruit peels were measured. Treating the fruits with 1-MCP better preserved the phytochemical compounds compared to simple refrigeration, preserving the fruit’s quality and prolonging its shelf-life. All the treatments help to maintain the glucose and fructose content and the acidity, preserving the aroma and organoleptic characteristics.
Hide AbstractImpact of Steam Extraction and Maceration Duration on Wines from Frozen ‘Frontenac’Must.
Svyantek, A., Wang, Z. & Hatterman-Valenti, H. (2023). Fermentation, 9(4), 317.
The enology industry in North Dakota is extremely young, with less than twenty years of existence. At times throughout the development of the North Dakota viticulture and enology industries, commercial wine producers have elected to purchase or store fresh harvested grapes as frozen musts. To investigate the fermentation outcomes related to skin contact for red grapevine musts, a postfreeze fermentation experiment was conducted with fruit from ‘Frontenac’, one of the most widely grown red grapevines in the Upper Midwest U.S. and North Dakota. Four fermentation treatments were applied to frozen ‘Frontenac’ grapevine musts: steam juice extraction, rosé, 1 day after inoculation (DAI) skin contact, and 9 DAI skin contact. Samples were collected daily for ten days and analyzed for fermentation progress and spectrophotometric monitoring of wine color attributes and total phenolics. The final wines were analyzed two years after bottling. Steam-extracted musts were initially darkest; however, they were lighter as final wines than the 9 DAI wines and similar to rosé wines in lightness. Total phenolics were greatest for 9 DAI wines and total red pigments were lowest for steam-extracted wines. While differences between treatments were detected, the wines remained visually similar; this indicates that color extraction within the freeze–thaw processes of musts may obliterate subtly and make it difficult to produce wines of light color when stored under these conditions. Continued work with additional grapevines beyond ‘Frontenac’ may help fine-tune must and fermentation extraction procedures for small-scale wineries growing cold-hardy grapevines.
Hide AbstractAgaricus bisporus chitosan influences the concentrations of caftaric acid and furan-derived compounds in Pinot noir juice and base wine.
Mederios, J., Xu, S., Pickering, G. & Kemp, B. (2023). Oeno One, 57(3), 255-268.
Chitosan is a fining agent used in winemaking, although its use in juice and wine beyond fining has been limited until now. Therefore, this study's first aim was to determine if chitosan derived from Agaricus bisporus (button mushrooms) could reduce caffeic and caftaric acid concentrations in Pinot noir grape juice (Study A). The second aim was to determine if chitosan, when added to base wine, could influence the synthesis of furan-derived compounds during storage (Study B). In Study A, Pinot noir grape juice was stored at 10°C for 18 hours after the following treatments: control (no addition), bentonite/activated charcoal (BAC), low molecular weight (< 3 kDa; LMW) chitosan, med. MW (250 kDa; MMW) chitosan, and high MW (422 kDa; HMW) chitosan (all 1 g/L additions). Caftaric acid was decreased, and total amino acid concentration was increased in the LMW chitosan-treated juice, while the estimated total hydroxycinnamic acid content, turbidity, and browning were decreased in the MMW chitosan-treated juice compared to the control. In Study B, Pinot noir base wine destined for sparkling wine was stored at 15 and 30°C for 90 days with the following treatments: control (no addition), LMW chitosan, MMW chitosan, and HMW chitosan (all 1 g/L additions). The three chitosan treatments stored at 30°C had increased furfural, homofuraneol, and 5-methylfurfural formation in the base wine compared to the control. At 15°C, furfural and homofuraneol had greater concentrations in all chitosan-treated wines after 90 days of storage. Our results demonstrate the potential of mushroom-derived chitosan to remove caftaric acid from grape juice and suggest that chitosan can influence the synthesis of furan-derived compounds in wine after short-term storage.
Hide AbstractReintroducing Autochthonous Minor Grapevine Varieties to Improve Wine Quality and Viticulture Sustainability in a Climate Change Scenario.
Frioni, T., Romanini, E., Pagani, S., Del Zozzo, F., Lambri, M., Vercesi, A., Gatti, M., Poni, S. & Gabrielli, M. (2023). Australian Journal of Grape and Wine Research, 2023.
One of the major challenges that global warming poses to viticulture is the maintenance of adequate acidity at maturity in white grapes for sparkling winemaking. This issue arises from three main occurrences: (i) with higher temperatures, degradation of malic acid is significantly enhanced; (ii) with a general advancement in grapevine phenology, grape maturity may occur under suboptimal climatic conditions; and (iii) harvesting grapes at “traditional” dates results in overripe fruits for sparkling destinations. In this biennial work, we compared the fruit and must composition of a local, widely grown white grape variety in the Colli Piacentini area (cv. Ortrugo, ORT) with those of a minor autochthonous variety, namely, Barbesino (BRB). Furthermore, we compared the composition, aromatic, and sensory profiles of wines obtained from ORT and BRB grapes picked on the same date and, in addition, of a second Barbesino wine from late harvest (BRB-LH). ORT and BRB had a similar sugar accumulation dynamic, whereas BRB exhibited a delayed loss of titratable acidity. In more details, BRB had lower malic acid degradation rates when malate concentration was <9 g/L. As a result, with comparable yield and total soluble solid content (TSS) (~20°Brix), BRB had a higher berry titratable acidity and malic acid concentration at harvest than ORT. BRB wines showed the highest titratable acidity (TA), while ORT had the lowest TA and a higher pH, and as expected, BRB-LH had the highest pH and a lower TA than BRB although still higher than those of ORT wine. The aroma profiles of wines were mainly characterized by fermentative aromas, including esters, fatty acids, higher alcohols, and C6 compounds, and BRB-LH wines showed the highest concentration of higher alcohols, while the fermentative esters were higher in ORT wines. Panelists considered BRB significantly fresher and with bigger aroma intensity than ORT, confirming that the higher acidity detected in BRB musts is well preserved in final wines. Our work demonstrates that local minor varieties can be reconsidered in light of the new climate change-related issues impairing viticulture sustainability today. In particular, currently neglected cultivars could help preserve must acidity as compared to traditional varieties having early ripening, maintaining the links with terroir and local traditions at the same time.
Hide AbstractDosing oxygen from the early stages of white winemaking: Effect on oxidation–reduction potential, browning stability, volatile composition, and sensory properties.
Benucci, I., Cerreti, M. & Esti, M. (2024). Food Chemistry, 432, 137243.
This study investigated the effect of controlled oxygen addition during alcoholic fermentation (4 mg/L, at 2nd, 4th, 6th, 10th, 14th, 16th, 20th day) and aging on lees (2 mg/L, every 15 days for 3 months) in a stainless-steel tank for white wine production from a nearly neutral grape. Under less reductive conditions (Ox), alcoholic fermentation was completed in 27 days, whereas one extra week was required under more reductive conditions (no-Ox). The greatest amount of dissolved oxygen in Ox wine triggered the increase of redox potential (EH) starting from the end of alcoholic fermentation and throughout aging (169 and 150 mV in Ox and no-Ox wine, respectively), also improving the wine resistance to browning. Oxygen addition from the early stages of winemaking significantly modulated volatile composition and sensory attributes, which may contribute to the diversification of wine style.
Hide AbstractGrapevine plantlets respond to different monochromatic lights by tuning photosynthesis and carbon allocation.
Liu, M., Zhao, Y., Fan, P., Kong, J., Wang, Y., Xu, X., Xu, M., Wang, L., Li, S., Liang, Z., Duan, W. & Dai, Z. (2023). Horticulture Research, 10(9), uhad160.
The quality of planting materials is the foundation for productivity, longevity, and berry quality of perennial grapevines with a long lifespan. Manipulating the nursery light spectrum may speed up the production of healthy and high-quality planting vines but the underlying mechanisms remain elusive. Herein, the effects of different monochromatic lights (green, blue, and red) on grapevine growth, leaf photosynthesis, whole-plant carbon allocation, and transcriptome reprograming were investigated with white light as control. Results showed that blue and red lights were favorable for plantlet growth in comparison with white light. Blue light repressed excessive growth, significantly increased the maximum net photosynthetic rate (Pn) of leaves by 39.58% and leaf specific weight by 38.29%. Red light increased the dry weight of the stem by 53.60%, the starch content of the leaf by 53.63%, and the sucrose content of the stem by 230%. Green light reduced all photosynthetic indexes of the grape plantlet. Photosynthetic photon flux density (PPFD)/Ci–Pn curves indicated that blue light affected photosynthetic rate depending on the light intensity and CO2 concentration. RNA-seq analysis of different organs (leaf, stem, and root) revealed a systematic transcriptome remodeling and VvCOP1 (CONSTITUTIVELY PHOTOMORPHOGENIC 1), VvHY5 (ELONGATED HYPOCOTYL5), VvHYH (HY5 HOMOLOG), VvELIP (early light-induced protein) and VvPIF3 (PHYTOCHROME INTERACTING FACTOR 3) may play important roles in this shoot-to-root signaling. Furthermore, the correlation network between differential expression genes and physiological traits indicated that VvpsbS (photosystem II subunit S), Vvpsb28 (photosystem II subunit 28), VvHYH, VvSUS4 (sucrose synthase 4), and VvALDA (fructose-bisphosphate aldolase) were pertinent candidate genes in responses to different light qualities. Our results provide a foundation for optimizing the light recipe of grape plantlets and strengthen the understanding of light signaling and carbon metabolism under different monochromatic lights.
Hide AbstractBiocontrol using Torulaspora delbrueckii in Sequential Fermentation: New Insights into Low Sulfites Verdicchio Wines.
Canonico, L., Agarbati, A., Galli, E., Comitini, F. & Ciani, M. (2023). Foods, 12(15), 2899.
Torulaspora delbrueckii showed renewed interest in recent years in the fermentation of wine, for its biotechnological potential linked to the ability to enhance flavor and aroma and it probably is the non-Saccharomyces yeast currently widely used in winemaking. On the base of this, sequential fermentations with a selected native strain of T. delbrueckii (DiSVA 130) and low sulfite native strain of Saccharomyces cerevisiae (DiSVA 709) were carried out to establish their contribution in biocontrol and aroma profile. A first set trials, carried out in winery, were set up to establish the effect of the sulfur dioxide addition on pure and T. debrueckii/S. cerevisiae sequential fermentations. A second set of sequential fermentations without SO2 addition were conducted in the same conditions, to evaluate the biocontrol and aromatic effectiveness of the T. delbrueckii native strain and a commercial one. The effective biocontrol action of native T. delbrueckii inoculated in sequential fermentation was shown, indeed without SO2 addition the presence of native T. delbrueckii revealed an effective fungistatic action in the first two days of fermentation. Moreover, the native T. delbrueckii strain seems to have fermentative performances comparable to those of T. delbrueckii commercial strain showing a more evident biocontrol action (wild yeasts reduced by c.a. 1 Log at 2nd day) and its presence did not negatively affect S. cerevisiae fermentation activity. Finally, the combination of both native and commercial T. delbrueckii/S. cerevisiae trials led distinctive aromatic profile of wines with a significant enhancement of isoamyl acetate, phenyl ethyl acetate, supported by positive appreciations, from the tasters, for ripe and tropical fruits, citrus and balance. The whole results indicate that the proposed strain could be a potential biocontrol tool toward wild yeasts in the first phase of fermentation also contributing to improve and differentiate the final aroma wine.
Hide AbstractSynthesis of a novel isotopically labelled standard for quantification of γ-nonalactone in New Zealand Pinot noir via SIDA-SPE-GC-MS.
Miller, G. C., Barker, D., Pilkington, L. I. & Deed, R. C. (2023). Analytical and Bioanalytical Chemistry, 1-13.
γ-Nonalactone is a linear aliphatic lactone ubiquitous in wine, associated with coconut, sweet, and stone fruit aroma descriptors. Little research has been conducted looking at the importance of this compound to New Zealand (NZ) wine aroma. 2H213C2-γ-Nonalactone, a novel isotopologue of γ-nonalactone, was synthesised in this work for use in a stable isotope dilution assay (SIDA) for quantification of γ-nonalactone in NZ Pinot noir wines for the first time. Synthesis was carried out using heptaldehyde as the starting material, and 13C atoms and 2H atoms were introduced via Wittig olefination and deuterogenation steps, respectively. The suitability of this compound as an internal standard was demonstrated by spiking model wine at normal and elevated conditions during sample preparation, with subsequent analysis via mass spectrometry showing stability of 2H213C2-γ-nonalactone. A model wine calibration, with concentrations of γ-nonalactone from 0 to 100 µg L-1, was shown to have excellent linearity (R2 > 0.99), reproducibility (0.72%), and repeatability (0.38%). Twelve NZ Pinot noir wines, representative of a range of NZ Pinot noir-producing regions, prices, and vintages, were analysed by solid-phase extraction-gas chromatography-mass spectrometry (SPE-GC-MS). The concentrations of γ-nonalactone ranged from 8.3 to 22.5 µg L-1, the latter of which was close to the odour detection threshold of this compound. These findings provide a basis for further research into γ-nonalactone and its impact on NZ Pinot noir aroma and provide a robust method for the quantification of this compound in Pinot noir.
Hide AbstractMetschnikowia pulcherrima in Cold Clarification: Biocontrol Activity and Aroma Enhancement in Verdicchio Wine.
Agarbati, A., Canonico, L., Ciani, M. & Comitini, F. (2023). Fermentation, 9(3), 302.
Non-Saccharomyces wine yeasts are not only proposed to improve the sensory profile of wine but also for several distinctive promising features. Among them, biocontrol action at different steps of the wine production chain could be a suitable strategy to reduce the use of sulfur dioxide. In this work, the activity of a selected strain of Metschnikowia pulcherrima was evaluated as inoculum in cold clarification with the aim to reduce SO2 and improve the aromatic profile of the wine. Fermentation processes were carried out at the winery level for two consecutive vintages using a pied de cuve as the starter inoculum coming from indigenous Saccharomyces cerevisiae strains. M. pulcherrima revealed an effective bio-protectant action during the pre-fermentative stage even if the timely and appropriate starter inoculum in the two years permitted the effective control of wild yeasts during the fermentation also in the control trials. In general, the main oenological characters did not show differences if compared with an un-inoculated trial, while the inoculum of M. pucherrima in cold clarification determined an enhancement of ethyl hexanoate, isobutanol, acetaldehyde, and geraniol even if they are considered in different amounts for each year. Indeed, the analytical and sensory profiles of wines were also influenced by the vintage and variation pied the cuve population. Nonetheless, the overall results indicated that M. pulcherrima led to biocontrol action and an improvement of the aromatic and sensory profile of the wine.
Hide AbstractDesign of a Plant-Based Yogurt-Like Product Fortified with Hemp Flour: Formulation and Characterization.
Montemurro, M., Verni, M., Rizzello, C. G. & Pontonio, E. (2023). Foods, 12(3), 485.
Plant-based milk alternatives have gained massive popularity among consumers because of their sustainable production compared to bovine milk and because of meeting the nutritional requests of consumers affected by cow milk allergies and lactose intolerance. In this work, hemp flour, in a blend with rice flour, was used to design a novel lactose- and gluten-free yogurt-like (YL) product with suitable nutritional, functional, and sensory features. The growth and the acidification of three different lactic acid bacteria strains were monitored to better set up the biotechnological protocol for making the YL product. Hemp flour conferred the high fiber (circa 2.6 g/100 g), protein (circa 4 g/100 g), and mineral contents of the YL product, while fermentation by selected lactic acid bacteria increased the antioxidant properties (+8%) and the soluble fiber (+0.3 g/100 g), decreasing the predicted glycemic index (-10%). As demonstrated by the sensory analysis, the biotechnological process decreased the earthy flavor (typical of raw hemp flour) and increased the acidic and creamy sensory perceptions. Supplementation with natural clean-label vanilla powder and agave syrup was proposed to further decrease the astringent and bitter flavors. The evaluation of the starter survival and biochemical properties of the product under refrigerated conditions suggests an estimated shelf-life of 30 days. This work demonstrated that hemp flour might be used as a nutritional improver, while fermentation with a selected starter represents a sustainable and effective option for exploiting its potential.
Hide AbstractPhysicochemical, nutritional, and functional characterization of gluten-free ingredients and their impact on the bread texture.
Arora, K., Tlais, A. Z. A., Augustin, G., Grano, D., Filannino, P., Gobbetti, M. & Di Cagno, R. (2023). LWT, 177, 114566.
Despite the commercial availability of gluten-free (GF) products, numerous nutritional, sensory, and textural limitations have been brought to the attention of the baking industries. This study aimed at the characterization of four GF flours (pregelatinized rice, pearl millet, common buckwheat, and soy protein isolate) for their nutritional and functional properties. Protein and starch were the major components in soy protein isolate and pregelatinized rice, respectively, whereas buckwheat and millet contained the highest amount of dietary fiber. Free phenolic compounds and antioxidant activity were at the highest levels in buckwheat flour followed by soy protein isolate. Likewise, all investigated ingredients varied greatly in their physicochemical properties. Based on single-ingredient baked-model, the effect on the texture and volumetric profiles of bread was reported, distinguishing GF ingredients in four different clusters with different characteristics. Accordingly, the four GF ingredients were combined to create a composite GF bread with acceptable textural properties approaching to those of a typical wheat bread. These findings might be regarded as a basis to design further innovative recipes and combinations using these raw GF ingredients.
Hide AbstractOptimization of a simultaneous enzymatic hydrolysis to obtain a high-glucose slurry from bread waste.
Sigüenza-Andrés, T., Pando, V., Gómez, M. & Rodríguez-Nogales, J. M. (2022). Foods, 11(12), 1793.
Bread and bakery products are among the most discarded food products in the world. This work aims to investigate the potential use of wasted bread to obtain a high-glucose slurry. Simultaneous hydrolysis of wasted bread using α-amylase and glucoamylase was carried out performing liquefaction and saccharification at the same time. This process was compared with a traditional sequential hydrolysis. Temperature and pH conditions were optimized using a response surface design determining viscosity, reducing sugars and glucose concentration during the enzymatic processes. The optimal conditions of pH and temperature in the saccharification stage and the simultaneous hydrolysis were pretty similar. Results show that the slurry produced with simultaneous process had a similar glucose yield at 2 h, and at 4 h a yield higher than that obtained by the sequential method of 4 h and could reduce time and energy.
Hide AbstractScheffersomyces stipitis ability to valorize different residual biomasses for vitamin B9 production.
Mastella, L., Senatore, V., Beltrani, T. & Branduardi, P. (2022). Microbial Biotechnology.
Sugar beet pulp (SBP), sugar beet molasses (SBM) and unfermented grape marcs (UGM) represent important waste in the agro-food sector. If suitably pre-treated, hexose and pentose sugars can be released in high quantities and can subsequently be used by appropriate cell factories as growth media and for the production of (complex) biomolecules, accomplishing the growing demand for products obtained from sustainable resources. One example is vitamin B9 or folate, a B-complex vitamin currently produced by chemical synthesis, almost exclusively in the oxidized form of folic acid (FA). It is therefore desirable to develop novel competitive strategies for replacing its current fossil-based production with a sustainable bio-based process. In this study, we assessed the production of natural folate by the yeast Scheffersomyces stipitis, investigating SBM, SBP and UGM as potential growth media. Pre-treatment of SBM and SBP had previously been optimized in our laboratory; thus, here we focused only on UGM pre-treatment and hydrolysis strategies for the release of fermentable sugars. Then, we optimized the growth of S. stipitis on the three media formulated from those biomasses, working on inoculum pre-adaptation, oxygen availability and supplementation of necessary nutrients to support the microorganism. Folate production, measured with a microbiological assay, reached 188.2 ± 24.86 μg/L on SBM, 130.6 ± 1.34 μg/L on SBP and 101.9 ± 6.62 μg/L on UGM. Here, we demonstrate the flexibility of S. stipitis in utilizing different residual biomasses as growth media. Moreover, we assessed the production of folate from waste, and to the best of our knowledge, we obtained the highest production of folate from residual biomasses ever reported, providing the first indications for the future development of this microbial production process.
Hide Abstract