00:02 Introduction & Kit Description
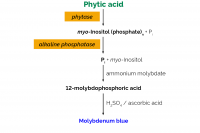
00:34 Principle
01:50 Preparation: (Solution A)
04:11 Preparation: (Solution B)
05:11 Preparation of Colour Reagent
05:56 Calibration
06:21 Assay Procedure: A. Sample Extraction
09:21 Assay Procedure: B. Enzymatic Dephosphorylation Reaction
14:35 Assay Procedure: C. Colourimetric Determination of Phosphorous
17:33 Calculations

50 assays per kit
Prices exclude VAT
Available for shipping
| Content: | 50 assays per kit |
| Shipping Temperature: | Ambient |
| Storage Temperature: | Short term stability: 2-8oC, Long term stability: See individual component labels |
| Stability: | > 2 years under recommended storage conditions |
| Analyte: | Phytic Acid, Phosphorus |
| Assay Format: | Spectrophotometer |
| Detection Method: | Absorbance |
| Wavelength (nm): | 655 |
| Signal Response: | Increase |
| Linear Range: | ~ 0.5 to ~ 7.5 µg of phosphorus per assay |
| Limit of Detection: | ~ 11.3 mg phosphorus (~ 40 mg phytic acid) |
| Reaction Time (min): | 25 min enzymic; 1 h for phosphate determination |
| Application examples: | Seed materials, feeds and foodstuffs. |
| Method recognition: | Novel method |
The Phytic Acid test kit is a simple method for the measurement and analysis of phytic acid/total phosphorus in food and feed samples. This method does not require purification of phytic acid via anion-exchange chromatography making it amenable to high numbers of samples.
Display our complete list of organic acid assay kits.

- Very cost effective
- All reagents stable for > 2 years after preparation
- Mega-Calc™ software tool is available from our website for hassle-free raw data processing
- Standard included
A Novel and Rapid Colorimetric Method for Measuring Total Phosphorus and Phytic Acid in Foods and Animal Feeds.
McKie, V. A. & McCleary, B. V. (2016). J. AOAC Int. , 99(3), 738-743.
Phytic acid, or myo-inositol hexakisphosphate, is the primary source of inositol and storage phosphorus in plant seeds and has considerable nutritional importance. In this form, phosphorus is unavailable for absorption by monogastric animals, and the strong chelating characteristic of phytic acid reduces the bioavailability of multivalent minerals such as iron, zinc, and calcium. Currently, there is no simple quantitative method for phytic acid; existing methods are complex, and the most commonly accepted method, AOAC Official MethodSM 986.11, has limitations. The aim of this work was to develop and validate a simple, high-throughput method for the measurement of total phosphorus and phytic acid in foods and animal feeds. The method described here involves acid extraction of phytic acid, followed by dephosphorylation with phytase and alkaline phosphatase. The phosphate released from phytic acid is measured using a modified colorimetric molybdenum blue assay and calculated as total phosphorus or phytic acid content of the original sample. The method was validated to a maximum linearity of 3.0 g phytic acid/100 g sample. Accuracy ranged from 98 to 105% using pure phytic acid and from 97 to 115% for spiked samples. Repeatability ranged from 0.81 to 2.32%, and intermediate precision was 2.27%.
Hide AbstractMorphological and nutritional assessment of Vigna vexillata (L.) A. Rich.: a potential tuberous legume of India.
Tripathi, K., Gore, P. G., Pandey, A., Nayar, E. R., Gayacharan, C., Pamarthi, R. K., Bhardwak, R. & Kumar, A. (2020). Genetic Resources and Crop Evolution, 1-12.
Vigna vexillate (L.) A. Rich. is a tropical tuberous legume which is fascinating for multiple uses in India. It is a climate-resilient legume and reported as a source of bruchid resistance, abiotic stresses tolerance and proteinaceous tubers. Meagre information is available for storage roots (tubers) of V. vexillate in general in the Indian scenario. In the present study, the evaluation of morphological and nutritional traits of storage roots are presented along with the genetic resources study. Herbarium study verified its wide distribution and occurrence in India since ancient time. Significant variability was observed for tuber morphological traits and nutritional parameters. Among accessions studied, IC259504 was identified promising agronomically and nutritionally both. Protein content in tubers of V. vexillate was recorded up to eightfold higher than that in sweet potato and tapioca. Results indicated that this species has the potential to meet the future needs of food and nutritional security and further utilization in Vigna improvement programme.
Hide AbstractThe silencing of TdIPK1 genes enhances micronutrient concentration in durum wheat grain.
Frittelli, A., Palombieri, S., Quagliata, G., Celletti, S., Astolfi, S., Botticella, E., Masci, S., De Vita, P., Volpato, M. & Sestili, F. (2023). Current Plant Biology, 35, 100309.
Phytic acid (PA) is the main storage form of phosphorus in kernel and is considered an anti-nutritional compound because of its ability to bind to essential minerals such as iron (Fe), zinc (Zn), potassium (K), calcium (Ca) and magnesium (Mg), thus limiting their availability, especially for populations whose diet is largely based on staple crops. This study reports a promising nutrient biofortification approach of durum wheat. The approach was based on the silencing of the gene encoding the inositol pentakisphosphate 2- kinase 1 (IPK1), involved in the last step of the PA biosynthetic pathway, through a Targeting Induced Local Lesions IN Genomes (TILLING) approach. Single knockout mutants for the IPK1 homeoalleles were identified and crossed to pyramid the two mutations. Although an elevated number of plants (F2 and F3 progenies) were analysed, no genotypes lacking both the homeoalleles were recovered, suggesting that the expression of IPK1 is crucial for seed formation in the spike and/or for plant germination and development. The characterization of the single null genotypes highlighted that the partial TdIPK1-B1- mutants showed a lower accumulation of PA in the kernel along with a higher content of essential microelements (Fe, Mn, Zn) compared to the control wild-type. The pattern of mineral accumulation was different for the TdIPK1-A1- mutants which only presented a greater accumulation of K.
Hide AbstractFood proteins from yeast-based precision fermentation: Simple purification of recombinant β-lactoglobulin using polyphosphate.
Hoppenreijs, L. J. G., Annibal, A., Vreeke, G. J. C., Boom, R. M. & Keppler, J. K. (2024). Food Research International, 176, 113801.
Proteins produced through precision fermentation are often purified through chromatographic methods. Faster and more cost-effective purification methods are desired for food application. Here, we present a simple method for purification of protein produced from yeast, using β-lactoglobulin secreted from Pichia pastoris as an example. The food-grade salt hexametaphosphate (HMP) was used to precipitate the protein at acidic pH, while the impurities (extracellular polysaccharides; mainly mannan) remained soluble. After re-solubilization of the protein-HMP complex by neutralization, excess HMP was selectively precipitated using calcium chloride. The protein content of the crude sample increased from 26 to 72 wt% (comparable to purification with anion exchange chromatography), containing only residual extracellular polysaccharides (9 wt%) and HMP (1 wt%). The established method had no significant impact on the structural and functional properties (i.e., ability to form emulsions) of the protein. The presented method shows potential for cost-effective purification of recombinant proteins produced through yeast-based expression systems.
Hide AbstractMinimal influence of milling technique in contrast to sourdough on the nutritional and organoleptic quality of bread.
Mietton, L., Mata-Orozco, J., Guezenec, S., Marlin, T., Samson, M. F., Canaguier, E., et al. (2024). Food Microbiology, 118, 104426.
A number of studies have shown the effect of cereals and sourdough on bread nutritional and organoleptic quality, but the impact of the milling technique remains little studied. There are two main types of milling technic depending on the bread-making food chain. Industrial bakeries mainly use roller mills while artisanal bakeries may also use stone mill. We set up a participatory experiment with six millers and four bakers to study the impact of these two milling techniques on the quality of flours, sourdough microbiota and the quality of breads. Millers made twenty-two different flours from four different wheat grain varieties using either roller or stone mills. Each baker initiated and maintained sourdoughs with three roller-milled and three stone-milled flours during at least 32 backsloppings and then made bread. The analysis of flours revealed a typical granulometry profile linked to wheat hardness with higher particle sizes for stone-milled flours. Stone-milled flours also had a higher maltose content. However, the milling technic did not drive the composition of the sourdough microbiota. Moreover, the analysis of bread revealed that variation in bread protein fractions and in bread aroma compounds were more related to the specific baker microbial community than to the milling technique. Carbohydrate contents were clearly linked to the main LAB species metabolism. These results revealed that the sourdough microbial community shapes the organoleptic and nutritional quality of bread more than milling techniques.
Hide AbstractMicrowave-assisted acidic hydrolysis of phytate from rye bran–Experimental procedure and model prediction.
Mayer, N., Dirauf, M. P. & Kaltschmitt, M. (2023). LWT, 189, 115499.
Phytate is an abundant phosphorus (P) compound in plant material, which cannot be taken up by monogastric animals but is to a significant part released into the environment with excreta. In order to make use of cereals' phytate content, phytate hydrolysis by microwave-assisted thermal treatment of an acidic rye bran extract into inorganic phosphate was investigated experimentally and by modelling for process application. A phytate conversion of 98.5% could be achieved at 200°C and 15 min reaction time and showed to be highly dependent on the temperature by a sigmoidal correlation. Treatment time showed its main influence in the mid temperature range (between 150 and 180°C), thus didn't play a role anymore at beginning hydrolysis and towards full conversion. A mathematical quartic model represented phytate conversion with an optimum at harshest examined conditions and allows for predictions of the hydrolysis within the parameter range investigated. A kinetic model gives parameters of reaction order 0.68 and an activation energy of 118.7 kJ/mol with the option to predict phytate hydrolysis beyond the investigated parameter range and for varying input concentration. Equal liberation of all six phosphate groups from phytate is mechanistically suggested. Both model approaches show comparable results.
Hide AbstractChemical and microstructural characterization of easy-and hard-to-cook Jack bean (Canavalia ensiformis (L.) DC.) collections.
Purwandari, F. A., Fogliano, V., de Ruijter, N. C. & Capuano, E. (2023). LWT, 189, 115451.
Differences in the microstructure and the chemical composition of pectic polymers were investigated from three Jack bean collections with different hard-to-cook (HTC) levels, as confirmed by the optimal cooking time analysis. These collections showed different cell and starch granule sizes, where the hardest-to-cook bean (Jombang collection) had the smallest maximum cell perimeter (285 μm), the smallest starch granule perimeter (98 μm), and the highest frequency of small intercellular space indicating more linkage in the middle lamella. No significant difference was observed in the frequency of small intercellular spaces between Malang and Cilacap despite having different optimal cooking times. Jack bean cotyledon cell walls were mainly composed of insoluble polysaccharide rich in arabinose and galactose. The beans contained high covalently bound pectin fractions (NEF) ranging from 78% to 83%. Higher bound phenolics (ferulic acid) were found with the increase in the optimal cooking times of the collections. Overall, the findings suggest the occurrence of cell wall linkage and highlight the importance of ferulic acid pectin crosslink in HTC Jack bean. This study could help develop strategies to reduce cooking time to promote the utilization of Jack bean.
Hide AbstractCharacterisation of physicochemical parameters and antibacterial properties of New Caledonian honeys.
Bucekova, M., Godocikova, J., Gueyte, R., Chambrey, C. & Majtan, J. (2023). Plos one, 18(10), e0293730.
Honey is an attractive natural product with various health benefits. A few honey-based commercial products have successfully been adopted in clinics to improve wound healing. However, screening of other potential sources of medical-grade honey, in particular, honeys from territories with high floral species diversity and high endemicity, is highly needed. The goal of this study was to characterise the physicochemical and antibacterial properties of New Caledonian honey samples (n = 33) and to elucidate the major mechanism of their antibacterial action. Inhibitory antibacterial activity of honeys against Staphylococcus aureus and Pseudomonas aeruginosa was determined with a minimum inhibitory concentration (MIC) assay. Enzymatic activity of glucose oxidase and the content of hydrogen peroxide (H2O2) in honey samples were analysed. Furthermore, total protein content of honeys together with their electrophoretic protein profiles were also determined in the study. The antibacterial efficacy of 24% of the tested honey samples was slightly superior to that of manuka honey with unique manuka factor 15+. The antibacterial activity of catalase-treated honey sample solutions was significantly reduced, suggesting that H2O2 is a key antibacterial compound of diluted honeys. However, the kinetic profiles of H2O2 production in most potent honeys at a MIC value of 6% was not uniform. Under the experimental conditions, we found that a H2O2 concentration of 150 μM in diluted honeys is a critical concentration for inhibiting the growth of S. aureus. In contrast, 150 μM H2O2 in artificial honey solution was not able to inhibit bacterial growth, suggesting a role of phytochemicals in the antibacterial activity of natural honey. In addition, the continuous generation of H2O2 in diluted honey demonstrated an ability to counteract additional bacteria in re-inoculation experiments. In conclusion, the tested New Caledonian honey samples showed strong antibacterial activity, primarily based on H2O2 action, and therefore represent a suitable source for medical-grade honey.
Hide AbstractExploitation of sprouted barley grains and flour through sourdough fermentation.
Perri, G., Minisci, A., Montemurro, M., Pontonio, E., Verni, M. & Rizzello, C. G. (2023). LWT, 187, 115326.
The local barley cultivar Nure was subjected to a controlled sprouting process, obtaining flour and grains that were fermented to produce type II sourdoughs. The germination process led to the reduction of starch and total dietary fibers (−58 and −48% compared to plain barley flour) and to the increase of phenolic compounds and protein bio-accessibility. Fermentation of sprouted barley (SB) with selected lactic acid bacteria determined a further enhancement of its nutritional features, by means of the increased free amino acids (up to 35%) and γ-aminobutyric acid concentrations (up to 57%), and decreased phytic acid content. The potential of SB sourdough to be used as ingredient in bread making was then investigated. Overall, the negative effects (on dough rheology and baking performance) related to the intense enzymatic activities characterizing the sprouted barley flour are strongly mitigated by the fermentation process. Therefore, besides improving bread nutritional and technological attributes, the use of SB sourdoughs, by supplying the native enzymes present in the sprouted grains, but in a less invasive form, could help decrease or substitute the use of commercial enzymes or flour improvers commonly used in the baking industry.
Hide AbstractDecorticated lentil malt flour: production process and use.
Cimini, A., Poliziani, A. & Moresi, M. (2023). Chemical Engineering Transactions, 102, 121-126.
In this work, the malting process of lentil seeds (Lens culinaris) was set-up to minimize their anti-nutrient content. The first (water steeping) and second (germination) process steps were studied in a 1.2-kg bench-top plant at 25°C. After 2-h steeping about 98.8% of seeds sprouted. As the germination process was prolonged for 72 h, the flatulence-inducing raffinose or phytic acid content was reduced by 94% or 63%, respectively. The third process step (kilning), carried out under fluent dry air at 50°C for 48 h and at 75°C for 3 h, gave rise to a gold metallic yellow-lentil malt, the cotyledons of which were cyclonically recovered and finally milled. The resulting decorticated yellow-lentil malt flour was used to prepare a fresh egg pasta high in raw protein (28±2 g/100 g), low in phytate (0.46±0.03 g/100 g) and in vitro glycemic index (38%), and approximately zero oligosaccharides.
Hide AbstractAssessment of oxalates and phytic acid contents in Asian green leafy vegetables: dietary recommendations.
Lee, H. W., Bi, X. & Henry, C. J. (2023). Food and Humanity, 1, 1223-1228.
Increased vegetable consumption is one of the recommended dietary interventions for optimal health but there is a lack of documentation about their anti-nutrients content, such as oxalates and phytic acid, which can modulate other health risks such as mineral deficiencies and urinary stone formation. The objective of this study was to evaluate the oxalates and phytic acid content of 26 green leafy vegetables (GLV) indigenous to and commonly consumed in Asia to expand existing nutritional information on food sources. The total oxalate contents ranged from 6 ± 0.2 mg/100 g fresh weight (FW) (iceberg lettuce) to 557 ± 47 mg/100 g FW (spinach), while the soluble oxalate contents ranged from 2 ± 0.3 mg/100 g FW (iceberg lettuce) to 501 ± 36 mg/100 g FW (turmeric leaves). The phytic content of the GLV ranged from 6 ± 2 mg/100 g FW (celery) to 111 ± 16 mg/100 g FW (moringa leaves). Overall, the anti-nutrients data have provided an increased understanding of these Asian GLV which will be important for nutritional and healthcare professionals to improve the dietary regime of the population. Data will also allow general consumers to make informed choices on how best these GLV can contribute to optimal health.
Hide AbstractPhysicochemical and chemical properties of mung bean protein isolate affected by the isolation procedure.
Wintersohle, C., Kracke, I., Ignatzy, L. M., Etzbach, L. & Schweiggert-Weisz, U. (2023). Current Research in Food Science, 7, 100582.
The effects of different mung bean protein isolation methods on the chemical composition, the physicochemical properties, and selected antinutritional factors of mung bean protein isolates were investigated. Six protein isolates were prepared by isoelectric precipitation at different extraction pH levels (pH 8 and 9), by micellization, and by hybrid isolation at varying salt concentrations (0.25 M, 0.50 M, 0.75 M). The extraction conditions affected the amount of antinutritive compounds of the isolates. Compared to mung bean flour, micellization reduced phytic acid content by approximately 48% and trypsin inhibitor activity by around 88%. The remaining phytic acid concentration of the isolates influenced their re-solubility, particularly under acidic conditions. The protein isolates exhibited significant differences in surface hydrophobicity and thermal characteristics, indicating structural modifications caused by the extraction methods. Micellization and extraction at pH 8 were identified as mildest isolation methods, as evidenced by the highest enthalpy values. SDS-PAGE analysis demonstrated an enrichment of globulins and comparable protein profiles among the isolates, suggesting that the observed differences arise from conformational changes rather than variations in protein composition. The product yield in protein extraction from mung beans ranged from 8% to 19%, emphasizing the importance of enhancing overall extraction efficiency or exploring the utilization of by-products obtained during the protein isolation process.
Hide AbstractComparison of Phosphorus and Phytase Activity Distribution in Wheat, Rye, Barley and Oats and Their Impact on a Potential Phytate Separation.
Natalie, M., Niklas, W. & Marvin, S. (2023), 16, 1076-1088.
When facing today’s scarcity of mineral phosphorus (P) resources and the environmental issues following enhanced P losses especially from agriculture, new solutions need to be implemented. In this framework, the potential for a mechanical separation of a P rich grain fraction from wheat, rye, barley and oats is investigated in order to provide animal feed with reduced organic P content. Thus, P accumulation in manure and soils should be prevented. Also, the subsequent utilization of the separated organic P, which occurs in the form of inositol P, for a sustainable P management via activation of intrinsic enzymes is evaluated. It was shown that in grain layers at 7.0, 5.5, 6.4 and 2.5% cross section of wheat, rye, barley and oats, respectively, maximum inositol P occurs with 1.6, 0.8, 1.4 and 1.2 g/100 g. Phytase activity is also highest in the outer layers of the grains with maxima of 9300, 12,000, 8400 and 2400 U/kg, respectively. A removal of the specific layers where inositol P is accumulated could possibly achieve a 24, 31, 60 and 27% organic P reduction for wheat, rye, barley and oats with 7, 14, 25 or 7% grain elimination. A debranning, eliminating all the outer grain layers to a certain extent, in contrast, leads to significantly higher mass losses. Within the P enriched layer determined from inositol P distribution, phytase activity is calculated to be around 285, 831, 777 and 42 U/kg for wheat, rye, barley and oats, respectively.
Hide AbstractExtrusion and drying temperatures enhance sensory profile and iron bioavailability of dry bean pasta.
Hooper, S. D., Bassett, A., Wiesinger, J. A., Glahn, R. P. & Cichy, K. A. (2023). Food Chemistry Advances, 3, 100422.
Bean flour is a highly nutritive, plant-based ingredient with the potential for great utility in many food products. Heat treated flours produced from commercial varieties of white kidney, yellow and black beans, were processed into pastas using high\low extrusion and drying temperatures. Bean pastas made with high extrusion\high drying temperature (H\H) had more favorable sensory attributes and better texture than those made with high extrusion\low drying temperature (H\L). Whereas bean pastas made with low extrusion\low drying temperature (L\L) were unacceptable. H\H pastas favored longer cooking time (8.6 – 13.8 min) versus those extruded at lower temperature (5.0 – 5.7 min). High extrusion temperature (100°C) with drying temperature high (90°C) improved iron bioavailability from yellow and white kidney bean pastas, 12.7 and 15 ng ferritin/mg protein, respectively as compared to black bean pasta (0.9 ng ferritin/mg protein). Cultivar, extrusion and drying temperatures are critical for producing bean pastas with high iron bioavailability.
Hide AbstractChemo-Enzymatically Modified Cereal Brans as a Modulator of Healthy Gut Bacteria: An In-Vitro and In-Vivo Study.
Devi, K., Maurya, R., Sharma, S., Bhadada, S. K., Bishnoi, M. & Kondepudi, K. K. (2023). Food Biotechnology, 37(3), 301-322.
Finger millet, kodo millet, and rice bran were chemo-enzymatically modified, and their prebiotic potential was evaluated using in-vitro human fecal batch fermentation and in-vivo feeding to mice. Modified cereal bran (mCB) from finger millet, kodo millet, and rice bran contained 30%, 17%, and 24% higher content of dietary fiber compared to their respective native bran. Tannin, flavonoids, polyphenols, and phytate content were reduced in the mCBs. Fecal batch fermentation with mCBs enhanced the short-chain fatty acid levels and the abundance of selected gut bacteria such as Bifidobacteria, Lactobacillus, and Bacteroidetes along with propionate and butyrate. In-vivo experiments showed no change in weight gain, blood glucose, and serum lipids. mCB supplementation enhanced the abundance of selected bacteria such as Bifidobacteria, Lactobacillus, and Bacteroidetes and decreased the abundance of E. coli, Klebsiella, and Clostridium. These findings provide an insight into the functional food potential of valorized mCBs in maintaining a healthy gut environment.
Hide Abstract